Meaning of pH
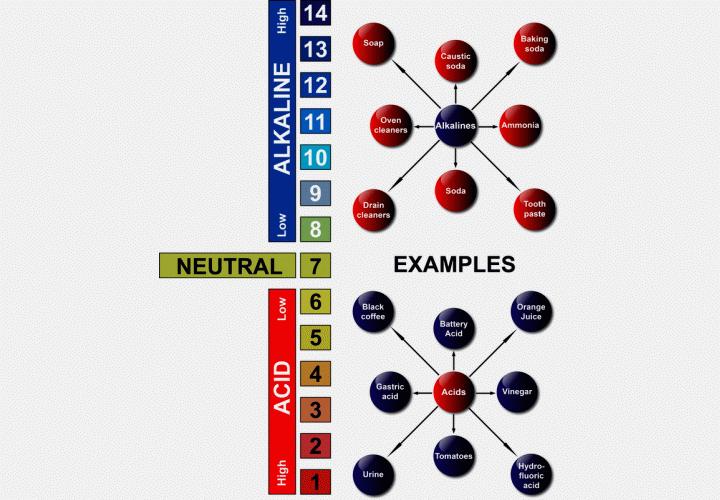
Q: I keep hearing about pH balanced shampoo? What does pH mean?
A: The term "pH" originates from the French term 'pouvoir hydrogen' (or Hydrogen Power) and has since been expressed as "parts hydrogen". pH refers to the number of hydrogen ions found in a substance. It's the amount of hydrogen ions present in a substance that determines the acidity or alkalinity of that substance.
Each whole number on the scale represents an increase or decrease in acidity or alkalinity by a factor of 10, thus, ammonia is over 1000 times more alkaline than baking soda, and lemon juice is almost 10 times more acidic than vinegar.
Alkalis and acids have different effects on the hair. Acids will harden and contract the hair, while alkalis expand and soften the hair shaft. Hair and skin have a pH level of between 4.5 and 5.5 (an average of 5), and any application of acidic and alkaline compounds can raise or lower the pH levels of the hair causing physical changes. Shampoos, conditioners and other chemical hair products use acids and alkalis to affect changes in the hair.
Many clarifying shampoos use alkaline ingredients to swell the hair shaft and allow the surfactants to penetrate more deeply. Most hair relaxers use a strong alkali to soften the hair, break the side bonds of the hair and remove curl. In addition, many conditioners and moisturizing shampoos use acidic compounds to contract the hair shaft, smoothing it and locking in moisture.
As an example of the effects of pH, consider the fact that distilled water which is pH neutral (7.0) is 100 times more alkaline than hair, and can swell the hair by as much as 20 percent.
©Hairfinder.com Image: Dreamstime
See also:
More about shampoo
The pH level of water in a hair salon
The pH level for hair dyes
What to look for when you need low pH shampoo and conditioner